In speaking with the C-suite team (CEO, CSO, and CMO) of MyoKardia ($MYOK) just mere months before their 13 billion dollar acquisition by Bristol Myers Squibb (BMS), and particularly after studying the company’s S-1, it became pretty clear to me that $MYOK credits and owes its success largely to a combination of savvy biochemistry (originally started in a Stanford University lab) followed up by building a robust precision medicine platform (see first few pages of S-1) to help design clinical trials that are focused in homogeneous, well-defined patient segments/populations. In my Zoom conversations with MyoKardia’s C-suite, I learned that the company employs whole-exome (not even whole-genome) sequencing to create targeted therapies that work best in heart disease patients with specific genomic profiles. Patient subpopulations are then stratified using bioinformatics data analysis approaches, with a focus on hypertrophic and dilated cardiomyopathy, including heart failure. In general, there is a critical unmet need for more and improved precision medicine approaches in cardiovascular/renal/metabolic (CVRM) drug discovery, but computational efforts to enable and sustain such innovation are just now emerging (some of them being developed in my own lab at the University of Chicago).
Given that BMS paid a 61% premium to acquire $MYOK, highlighting the scarcity of valuable assets in the cardiovascular disease space, it stands to reason that Sanofi must still be kicking themselves for walking away from MyoKardia last year in the roller coaster that is typical of cardiovascular disease drug discovery and clinical development. In my opinion, the high premium is also a consequence of Bristol Myers Squibb’s blockbuster drug, Eliquis (apixaban), coming off patent in several years despite attempts to stave off generic challengers for as long as legally possible. In other words, given MyoKardia’s FDA-approved mavacamten for symptomatic obstructive hypertrophic cardiomyopathy (HCM), and MyoKardia’s other promising drug programs (e.g., danicamtiv) in other clinical indications (e.g., dilated cardiomyopathy, heart failure, non-obstructive and other diseases of diastolic dysfunction), BMS is hoping to re-fill its cardiovascular drug pipeline.
And re-fill they will
My professional assessment is that BMS will have no problem re-filling its cardiovascular pipeline after Eliquis, maybe even with mavacamten alone… e.g., at least with respect to obstructive HCM, where the current standard of care is to use decades-old beta blockers or open heart procedures (surgery). But beta blockers are known to worsen diastole and do not actually help with the underlying disease progression characteristic of HCM. Since HCM has a progressive nature, beta blockers do not address the underlying disease pathology and, so while they may mask or inconsistently for some patients alleviate certain symptoms, the disease continues to progress in the background. The recognition of this phenomenon and helping physicians understand at what point their patients start increasing in their risk (and thus when they should try mavacamten) will likely be a significant future catalyst for boosting BMS stock price.
Eventually, I predict that mavacamten will be a therapy that is used more and more as a first line treatment, or used on top of (or in addition to) beta blockers, e.g., 90% of patients in the EXPLORER-HCM trial were already on a beta blocker or calcium channel blocker, with very positive results (addressing underlying hypercontractility, alleviation of obstruction, clean safety profile, etc.). Mavacamten may also be clinically useful to people who have moved off of beta blockers because they don’t tolerate them or because beta blockers don’t help alleviate their symptoms of obstruction. We may even see combination therapies of mavacamten being used alongside beta blockers or calcium channel blockers. So, taken together, my assessment is a strong net positive for BMS with respect to seeing long-term returns on their 61% premium.
What’s ahead?
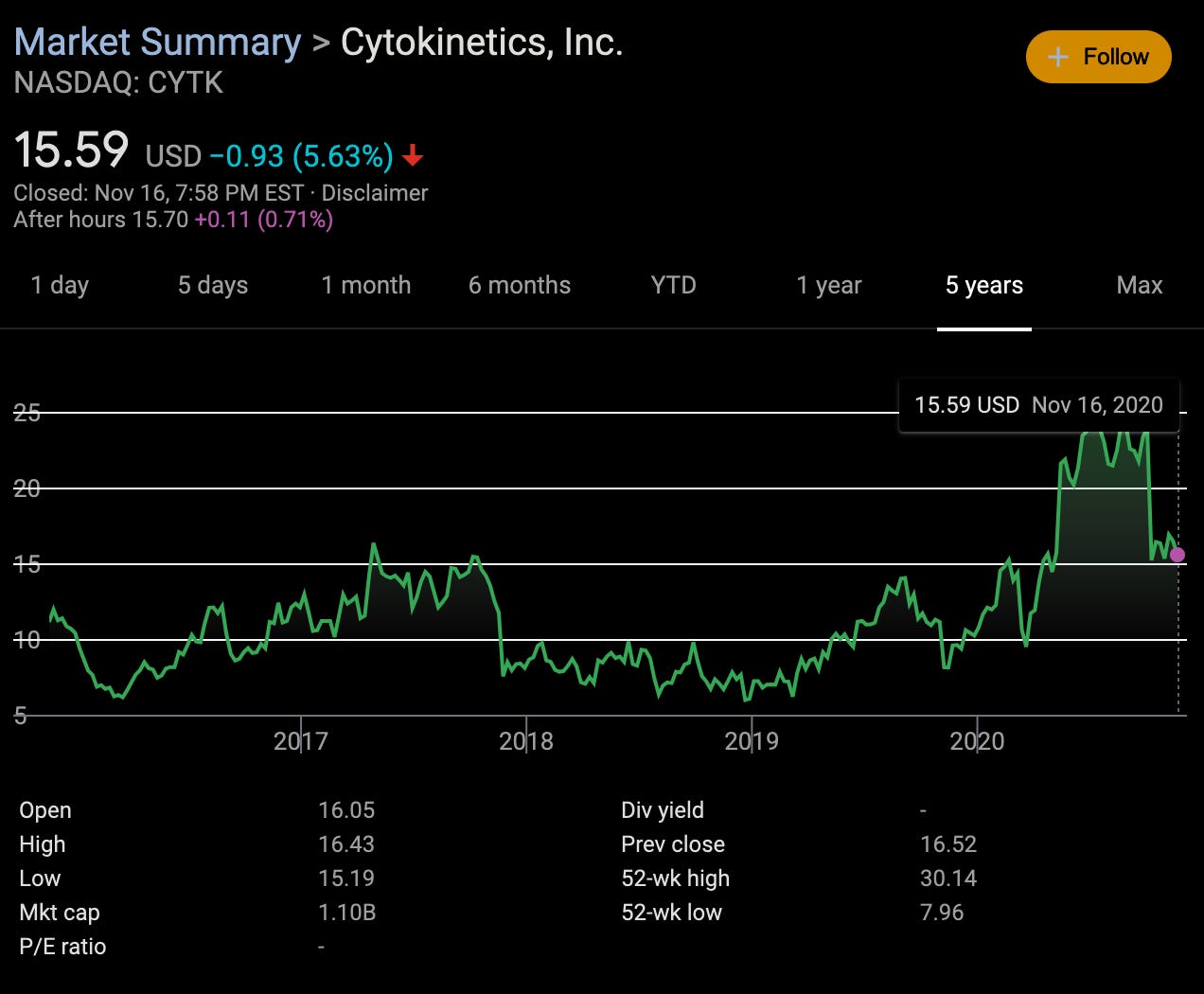
In my opinion, the only thing BMS would need to be worried about is potential competition, as Cytokinetics is developing a similar type of drug to mavacamten that also targets cardiac myosin and looks to be long-term hot on mavacamten’s heels with CK-274 and CK-271 (despite not being as far along in clinical development yet). Similar to the Sanofi/MyoKardia roller coaster drama highlighted earlier, Cytokinetics recently had its own roller coaster:
Specifically, the recent sharp decline is due to Amgen/Cytokinetics’ omecamtiv mecabril not meeting its secondary endpoint of reducing cardiovascular death (despite meeting the primary endpoint of reducing hospitalization) in the GALACTIC-HF trial. Heart failure (HF) is the leading cause of hospitalization in people 65 and older. What’s interesting is that despite recent good news out of Cytokinetics showing that omecamtiv mecabril works twice as well in subgroups of more severe HF patients with lower ejection fraction, investors are still skeptical despite a strong buy rating. From a scientific point of view, it is well-known that HF exhibits significant genetic heterogeneity, thus requiring precision medicine approaches to subphenotype HF patients into finer, more granular and precise subcategories of disease (i.e., not every HF patient is like any other HF patient, in fact quite different, but we don’t yet understand why and at what scale these differences in disease pathophysiology manifest themselves robustly). This highlights the need for more and improved precision medicine approaches, as described earlier. The feds are even organizing national competitions to address the heterogeneity inherent to heart failure diagnoses in an effort to subphenotype the disease better using precision medicine big data approaches. In my opinion, Cytokinetics shares should have climbed following the AHA news of the drug’s success in certain subgroups of HF patients but, as discussed in a previous post, because retail/generalist investors on Wall St. typically don’t follow the CVRM scientific thread this closely (at least with respect to following relevant NIH competitions etc.), I’m not surprised that shares dropped another 5%. But if there’s a time to buy low, now is a pretty good time and I think the strong buy rating is wholly justified.
In perspective, I see history repeating itself time and time again in the roller coaster of CVRM drug discovery and clinical development, as similar concerns revolved around MyoKardia at the time when Sanofi permanently disengaged in 2019. It’s just par for the course in this sector. But if the only thing you believe in is a strong co-founder team, you should know that one of the founders (James Spudich) who co-founded MyoKardia is also a co-founder of Cytokinetics. The science here is solid and the biotech market will naturally make its own corrections over time, while the prospects of precision phenotyping HF continue to improve.
If you enjoyed this post, sign up now so you don’t miss the next issue:
In the meantime, tell your friends!
P.S. Not investment advice. Read the Liability/Disclaimer subsection to learn more.




I am loving your blog! I did my post-doc on the Mas receptor and it’s great to learn about what people are doing in patient segementation in the cardiovascular space. I’m following right alongside In The Pipeline and Bruce booth. Please keep the posts coming.